
[ad_1]
The Digital Therapeutics Alliance defines a digital therapeutic (DTx) as “evidence-based therapeutic interventions which are pushed by high-quality software program packages to stop, handle, or deal with a medical dysfunction or illness.” One key query is what components do US payers bear in mind when evaluating DTx and the way does that differ from customary prescribed drugs. A paper by Gomez Lumbreras et al. (2024) held digital focus teams with 21 US payers to seek out the reply. Key concerns embrace:
- Want for Proof. Virtually all survey respondents (n = 19/21 90%) indicated they might require a scientific trial to think about protection of the product. This proof contains knowledge on efficacy, effectiveness and worth (together with cost-effectiveness perspective)
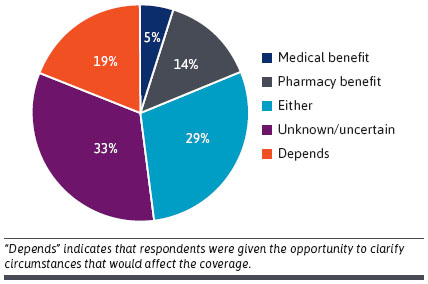
- DTx Protection: Medical, Pharmacy or Different? Many respondents had been unsure if reimbursement ought to undergo medical or pharmacy advantages. The bulk thought it will most likely be the pharmacy and therapeutics committee (n = 15/21 71%), however , a number of members answered “different” (n = 6/21, 29%) [see Figure below]
- FDA Regulation and Pending Laws. Total, 14/21 (66.7%) respondents would require an FDA analysis of the DTx product for it to be thought of for protection (particularly if lined beneath the pharmacy profit). Different respondents indicated that FDA analysis was helpful however not at all times required for protection. A number of payers cited the necessity for proof past the necessities of the FDA to think about a DTx product for protection (e.g., effeciveness, worth).
- Reimbursement: NDC vs. CPT. A prescription could be needed for a lot of well being plans to reimburse a DTx product on condition that many insurance policies exclude reimbursement for over-the-counter merchandise. Contributors extensively agreed {that a} coding system could be required, and {that a} Present Procedural Terminology (CPT) code or Nationwide Drug Code (NDC) could be probably the most environment friendly methods to make sure reimbursement.
- Boundaries. Boundaries talked about embrace sturdiness of remedy impact, value of merchandise, and mechanisms for reimbursement/cost. Different points included the position of DTx merchandise on affected person engagement and remedy adherence. Many perceived that DTx weren’t “bona fide” therapies partially as a result of some thought of them simply “apps” and comparable variations could possibly be downloaded on-line free of charge.
- Payer Administration. Some claimed that utilization administration insurance policies (e..g, prior authorization, step edits, amount limits) could possibly be used for DTx simply as they’re for pharmaceuticals. Others advised {that a} DTx product could possibly be a part of a care administration program fairly than masking it individually. A number of members defined that their organizations had been at the moment masking DTx merchandise as a part of scientific packages.
You’ll be able to learn the complete article with useful quotations right here.
[ad_2]